|
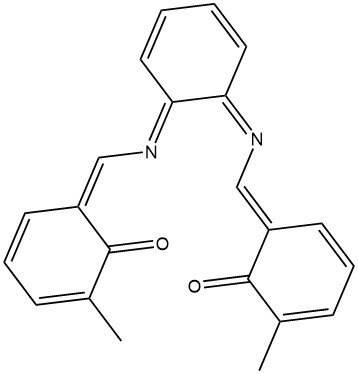
Wrong Lewis structure for CZM |
Correct Lewis structure for CZM |
 |
 |
PDB entry 1J3F is an artificial metallo-protein that contains a chromium ion, Cr(III), bonded to a CZM ligand. Both the metal ion and the ligand cause problems with MOZYME. The Cr3+ ion bonds to CZM-2 and to His-93 and H2O-203. This would give the system a net charge of +1, but because MOZYME does not have UHF capability, the oxidation state used for the chromium is Cr2+; this gives the system a net charge of zero. Using Cr2+ is obviously unrealistic, but until a UHF version of MOZYME is written, this is the best approximation available.
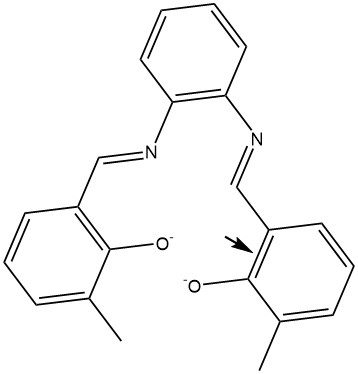
A more interesting problem is presented by the CZM ligand, residue: CZM-180. At the start of a MOZYME job the Lewis structure is calculated, in the case of CZM there are two possible Lewis structures, the one in the figure on the left is incorrect, the correct structure is on the right. Which of these would be chosen by the Lewis structure generator would depend on subtle clues. Uncertainty of this type can easily ruin a research program. Fortunately, to prevent the incorrect Lewis structure from being selected is straightforward. All that needs to be done is to define one of the C-C bonds adjacent to the oxygen atom in the phenyl ring to be a double-bond, as indicated by the arrow in the correct structure below.
|
Wrong Lewis structure for CZM |
Correct Lewis structure for CZM |
 |
 |
In the data-set for this protein the double bond is defined in two steps. First, keyword SETPI indicates that π-bonds are to be defined, then at the end of the data-set, the π-bond is defined. Both changes, the keyword and the extra data, can be seen in the ARC files for the various types of calculation.
If the Lewis structure generator would have made the correct choice of π-bonding, then the extra data would be redundant, but if there was a possibility of it making the wrong choice, the keyword SETPI would have prevented that from occurring.
Finally, this illustrates the limit of automated Lewis structure generation. An informed chemist would know which option to chose, but putting that information into code would be difficult - both structures look reasonable from one perspective or another.